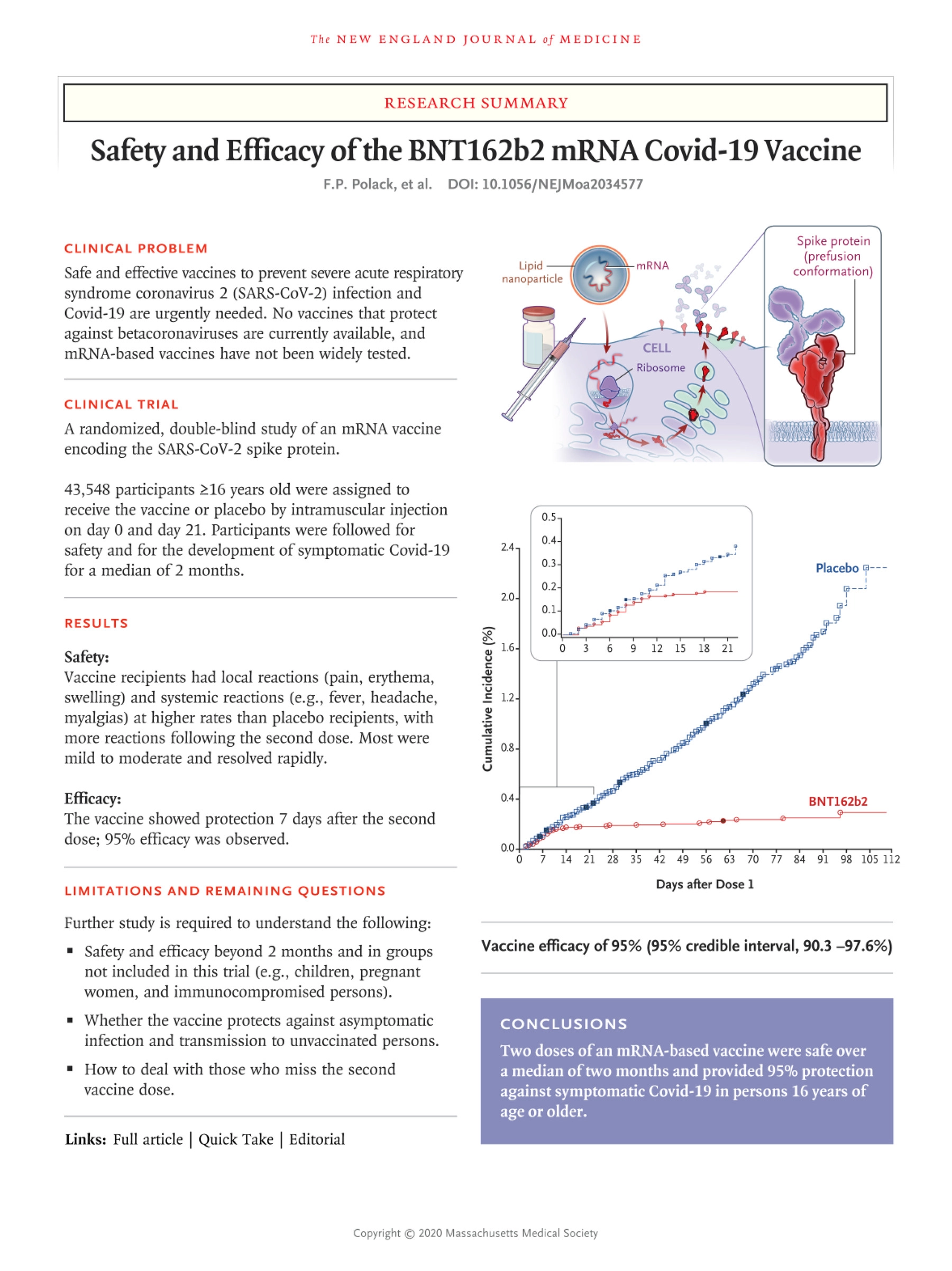
Six months safety and efficacy of Pfizer
Research funded by pharmaceutical companies claims vaccine safety
Leading medical journals have published many articles, that allegedly confirm the benefits of vaccination.
One typical example is a recently published preprint reviewing six months safety and efficacy of the BNT162b2 Pfizer vaccine. As it was funded by Pfizer/Biontech, involved mostly Pfizer employees, and lists a Pfizer employee as the corresponding author, the study can certainly not be called impartial.
https://www.medrxiv.org/content/10.1101/2021.07.28.21261159v1
Contrary to established scientific norms, the study was not double-blinded and, starting December 2020, participants were given the option of ‘unblinding’. Those in the Placebo group were offered the Pfizer shot. As a consequence, the actual follow-up period was much shorter than the six months indicated in the title (49% had <4 months post second dose), and no long-term study can be carried out in the future, due to the absence of a Placebo group.
The paper admits, that there were “imbalances in adverse events (30% vs 14%), related adverse events (24% vs 6%), and severe adverse events (1.2% vs 0.7%) between BNT162b2 and placebo groups”. However, it insists that “few participants had serious adverse events”.
As far as deaths from all causes are concerned, 15 Pfizer and 14 Placebo participants died. That the Pfizer group had one more death than the Placebo group can be a coincidence. Nevertheless, for a disease that is supposedly as deadly as Covid, we would have expected a lot more deaths among the unvaccinated than the vaccinated. It is interesting, that the most frequent cause of death was cardiac arrest, which affected 4 Pfizer participants, but only 1 member of the Placebo group.
Health authorities and politicians often refer to scientific papers, that certify high vaccine efficacy. Unfortunately, most of these studies are financed by pharmaceutical companies and it is easy for them to increase efficacy to the desired level, for instance by choosing a favorable trial design and setting convenient endpoints. It is a scandal, that pharmaceutical companies can evaluate their own products, and publish the results in leading medical journals as content, and not as advertisement.
Even if Big Pharma does not directly finance a specific study, it can exert enough pressure on scientists, for instance by threatening to withdraw funding for other projects. To expect most research papers to be impartial is naïve. (01)
See full report on Live Beyond Borders for a lot more in-depth review and references: Covid vaccines are not as safe as we are told – Sept 30, 2021 (02)
Controversy surrounding the original Pfizer Trial
A recent published paper on the first Pfizer Vaccine Trial has FINALLY awoken some of the doctors who have been calling people “antivaxxers” or “conspiracy theorists” instead of taking more of an “investigative journalistic” approach and checking their claims. See this post for full details about the original Pfizer Trial. (03)
BMJ: Unclean Data in Vaccine Trials (Pfizer & FDA should be investigated & Rollout Halted) – November 10, 2021 (04)
Pfizer Posts:
- MIT Exclusive: Record-Level Data from 3 Countries [Kirsch]
- Mike Yeadon’s (censored) democide & digital ID warning to Croatia [Dec 2023]
- WHO pushes new “Revaccination” marketing campaign for Pregnant Women [4 Dec 2023]
- NZ C19-Vax Massacre Data [Liz Gunn]
- C19-Vax-Lies Chat (Epidemiologist & Toxicologist)
- Funny Schwab-Gates WEF Parody Vids
- mRNA – most dangerous platform released on mankind ~Toxicologist [Senate Hearing]
- Pfizer switched templates that could cause permanent genome changes to those injected & their offspring…[Senate Hearing]
- Vaxxed Death count Southern Hemisphere [New Paper]
- Dental anaesthetics graphene/nanotech
- [The Voice] Pfizer says Yes – Red Flag number?
- Australian Senators vs Pfizer
- The Great Awakening [Documentary]
- Free Books to Recruit (Brainwash) Your Children into Life-long Pharma Slavery!
- Following the $CIENCE? (Bookmarks)
- The Sequel to the Fall of the Cabal [Documentary]
- [Part II] Dr Melissa McCann on Pfizer, TGA, FDA, LongCovid/VAED & Excess Deaths
- [Must See!] Aussie GP – Dr Melissa McCann – Stands up [Part I]
- [Legal] Vax-Injured Class Action – Australia
- Dr Peter Hotez (Fraud-chi’s replacement)
- RIP † Dr Arne Burkhardt (Pathologist)
- [Legal] Bioweapon evidence [May 23, 2023]
- CDC & Media “Pandemic of the Unvaccinated” Lies #WeWontForget
- Covid Vaccines deliver 5G Nanotechnology (La Quinta Columna)
- Oz MSM | Then & Now | #WeWontForget
- [2e] Game is over: Magnetic phenomenon & The Graphene Flagship
- [2c] Game is over: “Graphene Oxide Corona” (a.k.a Spike Protein)
- [2a] Game is over: Graphene – Raman Evidence
- Conference: The Game is Over [1/6]
- [Canada] National Citizens Inquiry
- Vaccine/EMF-enabled Mind-Control [Hypothesis & Refs]
- Covid Mismanagement [Senator Roberts]
- Download Research
- BlueTRUTH [Part-1/5] Documentary
- Atrazine | Making Men more ‘Feminine’ (via Rain?)
- Pfizer – IBM – Collaborate – Internet of Things – 2017
- [ICIC] no new virus, no lab-leak, just a story & a bad test (Part 2 of 2)
- [ICIC] no new virus, no lab-leak, just a story & a bad test (Part 1 of 2)
- What your doctor won’t tell you about the Pfizer docs
- BioNTech Collaborators
- c19 Jabs causes multi-organ inflammation [Dr John Campbell]
- [Solutions] Serene Teffaha – CoVision
- Using ChatGPT to condemn Catholic Jobs requiring Covid Jabs
- RW 4/8.) OTA & Brook Jackson’s Pfizer Case
- [ICIC] Australian Aboriginals Fight for Survival
- WHO Pandemic Treaty Debate [Senator Gerard Rennick]
- More Videos on TGA FOI 2389 [Dr. John Campbell]
- TGA FOI 2389 – BigPharma & TGA Conflict$
- TGA FOI 2389 – Ingredients, Dosage, Manufacturing
- TGA FOI 2389 – No Benefit

Site Notifications/Chat:
- Telegram Post Updates @JourneyToABetterLife (channel)
- Telegram Chatroom @JourneyBetterLifeCHAT (say hi / share info)
- Gettr Post Updates @chesaus (like fakebook)
Videos:
References






![[Fauci] Media-Smear Campaign of Prominent Epidemiologists (Senate Hearing Jan 2022)](https://pennybutler.com/wp-content/uploads/2022/01/collins-to-fauci-quick-devastating-takedown.jpg)
