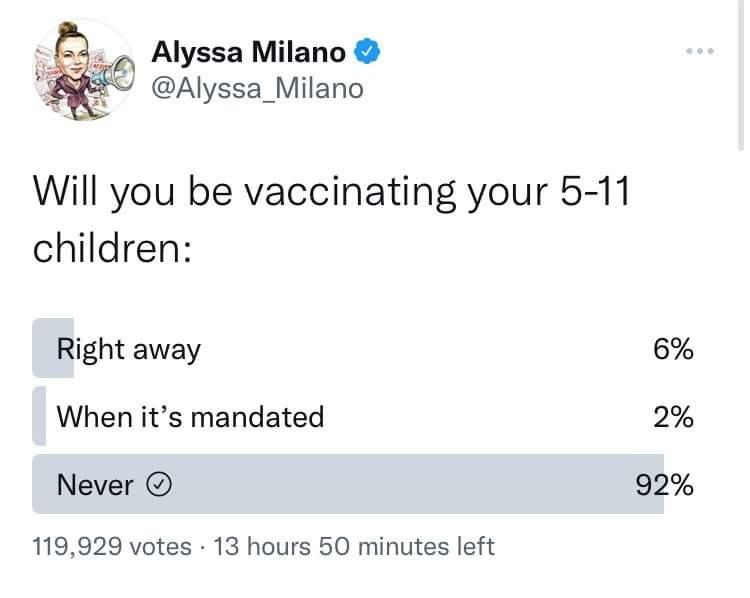
Pfizer Deaths – Kids (VAERS)

People pretending this isn’t happening? Humanity has gone? At what point will this be more real to those who deny it than the fear of something that is absolutely treatable without risking experimental things with completely unknown adverse effects – on kids – who had their whole life ahead of them – these are kids!
Random Summaries from Pfizer Deaths in Kids
(source: VAERS)
Fetus
The baby’s heart stopped beating.
A fetus patient received bnt162b2 (COMIRNATY), dose 1 transplacental on 12Aug2021 (Lot Number: FE7C1BSA) as dose 1, single for COVID-19 immunisation.
The mother pregnant in her 1st trimester, vaccinated on 12Aug2021 at 6 weeks pregnant.
On 19Aug2021 the parent had a miscarriage (metrorrhagia presenting symptom). The baby’s heart stopped beating. Though on her follow-up visit on 10Aug2021 the heart was beating. Ultrasound on 19Aug2021: interrupted pregnancy at 9 weeks. The baby died on an unspecified date.
12 Years Old
A 12-year-old Male patient received the second dose of Bnt162b2 (COMIRNATY) on 13Oct2021 (Lot/Batch: SDCN1) as 0.3 ml single dose for covid-19 immunisation. Found dead; Myocarditis; Headache; Fever; Appetite lost;
This is a spontaneous report from a non-contactable physician downloaded from the Regulatory Agency (A)-WEB, regulatory authority number DE-PEI-202100217297.
The patient’s medical history included Distal arthrogryposis, Cardiomyopathy. The patient previously received the first dose of bnt162b2 (COMIRNATY) on 22Sep2021 at the age of 12-year-old as 0.3 ml single dose for covid-19 vaccination/immunisation.
On 14Oct2021 the patient experienced Fever, Headache, Appetite lost.
On 15Oct2021 the patient experienced Found dead, Myocarditis.
Autopsy-determined Cause(s) of Death: Cardiac death in cardiomyopathy.
13 Years Old
A 13-year and 6-month-old male patient received second single dose of bnt162b2 (COMIRNATY: Solution for injection; Batch/Lot Number: FK0108; Expiration Date: 30Apr2022), via an unspecified route of administration on 30Oct2021 16:55, as dose 2, single for covid-19 immunisation.
Medical history included bronchial asthma from an unknown date and unknown if ongoing. Prior to vaccination, the patient was not diagnosed with COVID-19.
The patient previously received first single dose of BNT162b2 (COMIRNATY, Solution for injection, Lot number FJ1763, Expiration date 30Apr2022) via an unspecified route of administration on an unspecified date in Oct2021 as dose 1 ,single for COVID-19 immunization.
The onset date/time of the event was reported as 30Oct2021 at 21:20. On 30Oct 2021 at 21:20, (day of vaccination), the patient developed cardio-respiratory arrest and died.
The event required emergency room visit. On 30Oct2021 at around 21:00, about 4 hours after the second dose of the vaccination, the patient found submerged in the bathtub.
The course of the events was as follows:
On 30Oct2021, after receiving the second dose of the vaccination, the patient returned home. At around 19:00, the patient had a meal. At 20:30, he took a bath, but he did not get out of the bathroom. At around 21:20, his parents found that he was in cardio-respiratory arrest.
They called an ambulance and at 21:34, an emergency team arrived. At 21:40, they arrived at the reporting hospital where cardiopulmonary resuscitation was performed, but his death was confirmed.
The reporting physician classified the event as serious (fatal outcome) and assessed the causality between the event and BNT162b2 as assessable. From the findings from the macroscopic anatomy, no possible cause of the event was found.
The reporting pharmacist commented as follows:
The fatal case of COVID-19 vaccine was reported from the emergency medical care centre. The patient had past medical history of bronchial asthma, however, considering that he was a healthy child and suddenly died, the possibility of BNT162b2 being related to the adverse event was considered to be high. After his death was confirmed, an autopsy was performed by the police. The reporting physician commented as follows: On 02Nov2021, judicial autopsy was performed. From the results, the cause of drowning could not be macroscopically detected.
Cardio-respiratory arrest; the patient found submerged in the bathtub;
Reported Cause(s) of Death: Drowning; Cardio-respiratory arrest.
13 Years Old
A 13-years-old male patient received bnt162b2 (COMIRNATY, Formulation: Solution for Injection, Batch/Lot Number: 10020A) dose 2 0.3 mL via an unknown route of administration on 11Aug2021 as dose 1, single for COVID-19 immunisation.
The patient’s medical history included extremely preterm (less than 28 weeks), Hypoxic-ischaemic encephalopathy, post haemorrhagic hydrocephalus, symptomatic epilepsy, dysphagia, respiratory insufficiency, reflux oesophagitis on an unspecified date.
The patient previously received first dose of bnt162b2 (COMIRNATY, Formulation: Solution for Injection) dose 1, Batch/Lot number unknown) for COVID-19 immunisation on 16Jun2021. The patient underwent lab tests and procedures which included platelets min 3410x 31uml, Quick <10, INR$g4.9, pTT $g180 sec, proBNP 24937 pg/ml, Trop T 1580 pg/ml on an unknown date.
The patient experienced multiorgan failure, lung haemorrhage, disseminated intravascular coagulation, pyrexia and septic shock on 13Aug2021.
The patient died due to Unknown cause of death on 14Aug2021.
13 Years Old
Patient received first COVID vaccine on 7/11/21. She was well until 7/26/21 when she developed nausea, vomiting, shortness of breath and dizziness.
At approximately 04:00 on 7/27/21, she woke up feeling poorly, went into the bathroom and then lost consciousness.
EMS was called, she was in a tachycardia at 180 bpm, BP 88/64, unresponsive. EMS report states she was “in SVT and cardioverted at 30J then 70 J. Still in SVT but becoming more alert. HR 165 bpm, sats 98%.”
In ED, she was felt to be in ventricular tachycardia, She received attempted resuscitation with multiple rounds of CPR including chest compressions, defibrillation for reported ventricular fibrillation, and medications.
She was unable to be resuscitated and death was pronounced in the ED. Autopsy is pending.
14 Years Old
A 14-year-old male patient received bnt162b2 (COMIRNATY), 1st dose on 06Jul2021 (Lot Number: EX0893) and 2nd dose on 27Jul2021 (Lot Number: EW2246, both via an unspecified route of administration as single dose for COVID-19 immunization.
Medical history none.
This is a spontaneous report from a Pfizer-sponsored program, from a contactable consumer (patient’s uncle) through the Pfizer company doctor.
The patient experienced pulmonary edema (death) on 03Sep2021. The patient died on 03Sep2021. The autopsy revealed that the cause of death was pulmonary edema.; Reported Cause(s) of Death: pulmonary edema
15 Years Old
Found unconscious; breathing difficulties; autopsy was performed on the same day at 3:40pm and found there was spontaneous bronchospasm; he was pronounced dead on the same day;
A 15-year-old male patient received bnt162b2 (COMIRNATY), dose 2, on 23Oct2021, single for COVID-19 immunization, received at his school.
Patient also did not report any problems or complications after receiving the first dose injection.
The patient experienced injection site pain (non-serious) on 23Oct2021, breathing difficulties (hospitalization) on 24Oct2021 09:30, was found unconscious (hospitalization) on 24Oct2021 09:30, autopsy was performed on the same day at 3:40pm and found there was spontaneous bronchospasm (death) on 24Oct2021 10:40, he was pronounced dead on the same day (death) on 24Oct2021 10:40.
The patient was hospitalized for breathing difficulties and was found unconscious from 24Oct2021 for 1 day. Therapeutic measures were taken as a result of breathing difficulties, was found unconscious. The patient died on 24Oct2021. An autopsy was performed that revealed found there was spontaneous bronchospasm.
The clinical course of events was as follows:
He was reported to have had injection site pain only. However a day after he received his second jab, he was found to have suffered from breathing difficulties and then was found unconscious at his home at about 9.30am. Patient was rushed to the private hospital here but he was pronounced dead on the same day (24Oct2021) at 10.40am. An autopsy was performed on the same day at 3.40pm and found there was spontaneous bronchospasm. All tissue samples in the body were sent for further examination by a hospital’s pathology forensic physician. The autopsy report is categorised as still incomplete because there are still tests that have not been received.
Autopsy-determined Cause(s) of Death: found there was spontaneous bronchospasm
15 Years Old
A 15-year-old female patient received bnt162b2 (COMIRNATY), intramuscular on 11Jul2021 07:30 (Lot Number: Unknown) (at the age of 15-year-old) as dose 1, single for COVID-19 immunization.
This is a spontaneous report from a contactable physician downloaded from the Regulatory Authority-WEB [FR-AFSSAPS-MP20215396].
Medical history included ongoing asthma, ongoing Barlow’s syndrome, ongoing Marfan’s syndrome. The patient’s concomitant medications were not reported. In good health overall, apart from a loss of 10kg over one year (since entering high school).
During the day (11Jul2021), asthenia and isolated arm pain. The next day (12Jul2021), headaches yielding under Doliprane. On 13Jul2021, around 16:30 (last moment conscious view), her mother drops her off to her father.
Father watered the garden and she cleaned the garage to prepare for her birthday party.
On 13Jul2021 17:20, her father found her in cardio respiratory arrest, back to the ground, next to a ladder.
At 17:30 arrival of firefighters: 2 external electric shocks were given and 1 mg of adrenaline injected. Moderately reactive pupils. At 17:50 arrival of Specialist mobile emergency unit: asystole (Life-threatening). Two injections of 1 mg of adrenaline, transition to ventricular fibrillation. 2 external electric shock, 2 ampule of Cordarone and one ampule of Calcium Gluconate. Return to regular sinus rythme without disturbance of repolarization and resumption of a pulse. Orotracheal intubation (probe no 6). New: 1 external electric shock, one ampule of Cordarone and 1 mg of adrenaline. Return of a sinus rhythm but presence of a sub ST in infero lateral. 90/60 mmHg arterial pressure excluding sedation. Tight areactive bilateral miosis pupils. Ventilated in Ventilator-Associated Conditions but presence of spontaneous ventilation requiring sedation by Hypnovel and Sufentanyl and 10 mg of Nimbex. Parallel introduction of Noradrenaline 0.8 mg/h. No filling. In total: low flow of 30 minutes.
Recovered and transfer to intensive care.
Examinations: biology: complete blood count normal, C-reactive protein 1.4. Coroner considered as normal no coronary dissection. Computed tomography scan Computed tomography arterial portography: No aortic dissection or large vsx, no intracranial bleeding, the super sigmoid aortography does not show any aortic insufficiency. The ascending aorta is moderately dilated. Computerised tomogram head: no bleeding, no traumatic injury. Electrocardiogram: Not very evocative. Respiratory rate. Maintenance of sedation, temperature control at 36 degrees. Complicated cardiac arrest of a Takotsubo. Trans-thoracic echocardiography finding a 30% altered left ventricular ejection fraction with kinetic disorders suggestive of Takotsubo (post stress?). More doubt about intra-left ventricular thrombus. Low left ventricular filling pressures. Integral time speed= 8. Inferior vena cava= 15. 15Jul2021 Appearance in the morning of continual clonies of the multiple sulfatase deficiency, put under Keppra increased to 750×2. Electroencephalography results pending + Left transcranial doppler more disturbed than the right (Vdiastolic 20 vs 40 on the right), Control contrast enhanced computed tomography scan superimposable at the level of large vsx, but appearance of parenchymal parenchymal hemispherical hemispherical right upper cerebellar areas of ischemic appearance. 20Jul2021 pathological awakening, inhalation lung disease, myocarditis assessment in progress (negative).
23Jul2021 no sign of waking up flat electroencephalogram alternating with a few waves of intermittent activity. Computered tomography scan stability of ischemic lesions appearance of cerebral edema compatible with anoxo-ischemic lesions, put under Mannitol. Cardio: cardiac magnetic resonance imaging in favor of a takotsubo, myocarditis unlikely, infective and immunological workup negative. 27Jul2021 Pathological electroencephalogram, Keppra introduction. Computered tomography scan increase in cerebral edema reaching almost the entire sustentorial stage, sudden episodes of desaturation.
The COVID serology returns positive (Ig G antiS and antiN and IgM), re-reading of the entry serology concluded with a Covid infection starting at the same time as the anti-covid vaccination.
30Jul2021 retro-rolandic aspect of brain death, vegetative coma. Decision to limit therapy. Complete file no further information. The patient died on 07Aug2021. An autopsy was not performed. Cause of Death: Anoxia cerebral and Cardiac arrest while outcome of the other events was unknown. Reported Cause(s) of Death: Cardiac arrest; Anoxia cerebral.
15 Years Old
A 15-year-old male patient received the first dose of bnt162b2 (PFIZER-BIONTECH COVID-19 VACCINE), in the left arm on 09Aug2021 at 09:00 AM (lot number: FF2154) as DOSE 1, SINGLE for COVID-19 immunisation.
The event resulted in Emergency room/department or urgent care. Prior to vaccination, patient did not diagnose with COVID-19. Device date was 29Aug2021. No treatment received.
The father reported that the patient (the son) received the dose 1 of the Pfizer vaccine on 09Aug2021 at 9AM on left arm. Unfortunately, patient passed away (death) on Friday 13Aug2021 at 04:30 without any history of illness.
16 Years Old
A 16-year-old (also reported as 16-year and 6-month-old) male patient received bnt162b2 (COMIRNATY, Lot Number: FF0843; Expiration Date: 31Oct2021), dose 1 on 10Aug2021 13:00 as single dose for covid-19 immunisation.
This is a spontaneous report from a contactable physician received from the Regulatory Agency. Regulatory authority report number is v21126302.
Medical history was none. The patient had no family history. There were no points to be considered on the vaccine screening questionnaire (primary diseases, allergies, vaccinations and illnesses within the last one month, medications the patient was taking, past adverse effect history, growth status).
Body temperature before vaccination was 36.2 degrees Centigrade.
On 10Aug2021 at 13:00 (the day of vaccination), the patient received the first dose of BNT162b2.
On 16Aug2021 at 16:40 (6 days, 3 hours and 40 minutes after the vaccination), the patient experienced cardiac arrest.
The patient was immediately transported and admitted to the reporting hospital. On 01Sep2021 (22 days after the vaccination), the outcome of the event was fatal.
The course of the event was as follows:
On 16Aug2021 at 16:40 (6 days, 3 hours and 40 minutes after the vaccination), the patient was immediately transported the reporting hospital because of cardiopulmonary arrest (CPA). Before arrival to the hospital, automatic external defibrillator (AED) was conducted once. After the arrival, asystole, then VF(ventricular fibrillation) were noted. And return of spontaneous circulation (ROSC) was achieved after the one attempt of direct-current defibrillator (DC). The patient was hospitalized after resuscitated from the CPA. An obvious cause of cardiac arrest was unknown on computerised tomogram (CT), the blood test and electrocardiogram. The patient underwent tracheal intubation, and was managed by artificial respiration under hospitalization. Although targeted temperature management (TTM) was performed, there was no recovery of consciousness.
On 31Aug2021 (21 days after the vaccination), blood pressure decreased. NOMI(Non-occlusive mesenteric ischaemia) was diagnosed through CT. On 01Sep2021 (22 days after the vaccination), the death was confirmed. The reporting physician classified the event as serious (Hospitalized) and assessed that the causality between the event and BNT162b2 as unassessable. Other possible cause of the event such as any other diseases was not reported.
The patient died on 01Sep2021. It was not reported if an autopsy was performed. The outcome of event ventricular fibrillation was recovered on 16Aug2021, the rest of events was fatal.;
Reported Cause(s) of Death: Cardiac arrest; cardiopulmonary arrest; Non-occlusive mesenteric ischaemia
16 Years Old
Was very tired and had sore muscles on September 09. Found September 10, 5:25 am dead.
There were excessive amounts of blood along with large blood clots that appear to have come vaginally.
16 Years Old
A 16-years-old female patient received BNT162B2 (PFIZER-BIONTECH COVID-19 VACCINE), as DOSE 2, SINGLE at the age of 16-year-old for covid-19 immunisation.
Patient previously received the first dose of BNT162B2 (PFIZER-BIONTECH COVID-19 VACCINE) on 13Jul2021 at the age of 16-year-old for covid-19 immunization. The patient experienced death on 13Oct2021.
Reported Cause(s) of Death: death
17 Years Old
Patient reported symptomatic (non-severe) case of COVID-19 August 2021 and recovered fully. She reported receiving Pfizer COVID vaccine 9/3/21 and second dose 9/15/21.
She present to the emergency department of my hospital 10/23/21 with chest pain and dyspnea for 48h. Was feeling completely well prior to onset of chest discomfort. Symptoms were mild. No sick contacts or family members.
ED evaluation remarkable for normal exam, no hypoxia, normal blood pressure. EKG with diffuse ST elevation. Troponin elevated at 20. CTA chest negative for PE or pneumonia. SARS-CoV-PCR positive but thought to be persistent positive rather than reinfection because of lack of clinical symptoms, recent COVID-19 and recent vaccination.
Cardiologist consulted, thought acute coronary syndrome unlikely based on age and lack of risk factors. STAT Echo resulted depressed EF 40-45%. Simultaneously she had become increasingly tachycardic and EKG appeared more ischemic. Cardiac cath lab was activated and she was about to be transported when she suffered cardiac arrest. Initial rhythm was VT. Received ACLS protocol CPR x 65 minutes including multiple cardioversion, amiodarone, lidocaine, magnesium and other antiarrhythmics.
Unfortunately she was not able to be resuscitated and died.
Cause of death possible acute myocarditis.
17 Years Old
A female patient of 17-years-old received first dose of bnt162b2 (COMIRNATY) as DOSE 1, SINGLE for covid-19 immunisation.
Medical history included Raynaud’s phenomenon from an unknown date and unknown if ongoing. Prior to the vaccination, the patient was not diagnosed with COVID-19.
The reporter reported an adverse event that occurred in the school where she worked, a student took the 1st dose of the pfizer vaccine and she had a reaction and was hospitalized in serious condition in the hospital. The reporter informed that the patient had died on an unspecified date for an unspecified reason.
Reported Cause(s) of Death: had a reaction and is hospitalized in serious condition / the patient had died
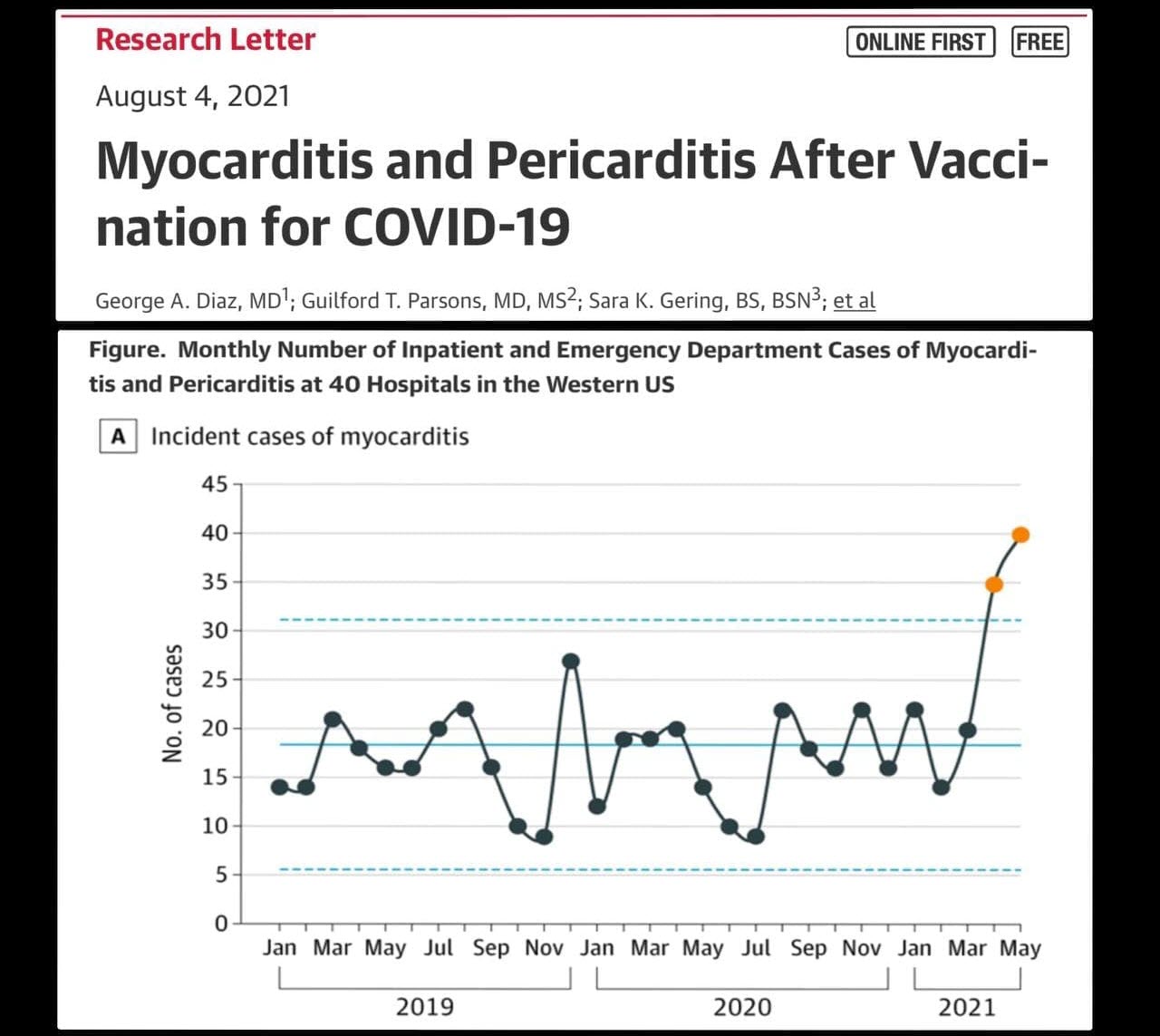
CDC’s Advisory Committee to add warning statement for Heart Inflammation in Adolescents

The Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) said there is a “likely association” of “mild” heart inflammation in adolescents and young adults after vaccination with an mRNA COVID vaccine and a warning statement is warranted.

According to a report by the COVID-19 Vaccine Safety Technical (VaST) Work Group, the risk of myocarditis or pericarditis following vaccination with the mRNA-based shots in adolescents and young adults is notably higher after the second dose and in males.
There have been more than 1,200 cases of myocarditis or pericarditis in 16- to-24-year-olds who received an mRNA COVID vaccine, according to a series of slide presentations published at the ACIP meeting.
Myocarditis is inflammation of the heart muscle that can lead to cardiac arrhythmia and death. According to researchers at the National Organization for Rare Disorders, myocarditis can result from infections, but “more commonly the myocarditis is a result of the body’s immune reaction to the initial heart damage.”
Pericarditis is often used interchangeably with myocarditis and refers to inflammation of the pericardium, the thin sac surrounding the heart.
Do not get your education from the media – do the research!
If you can’t find the legitimate research that isn’t being shared on the nightly news or social media as it may cause “vaccine hesitancy” (which is apparently a crime these days due to the nefarious upside down world we’re in right now where evil has the megaphone!), start by downloading mine above, or browsing this site and others like it.
Please, I beg you.




Posts tagged “CovidKids”

Free Books to Recruit (Brainwash) Your Children into Life-long Pharma Slavery!
Found the mother-load of free propaganda books for kids via the Pfizer Clinical Trial site!

[Canada] National Citizens Inquiry
A must-watch heart-breaking inquiry where Canadians testify on how the covid policies affected their lives.
Download Research
Everything I’ve saved so far in my quest to get answers during the pandemic. Truth is all I seek.

Vaccine Generated Spike in the Blood of Children with Myocarditis
Deep dive and brief dive into the new study proving vaccine-generated spike protein in the blood of children that were diagnosed with Myocarditis.

TGA FOI 2389 – BigPharma & TGA Conflict$
Redacted information. Bureaucracy. Vaccine injuries. Ignoring patient data. TGA Conflict$ with BigPharma. ATAGI’s admission about myocarditis. Novel technology on the world instead of tried & true. Multiple agencies passing the buck to each other. Doubling-Down in Omicron instead of easing off. Mandating injections for Wuhan strain in Omicron. Australia’s deal with Moderna. mRNA flu shots. More.
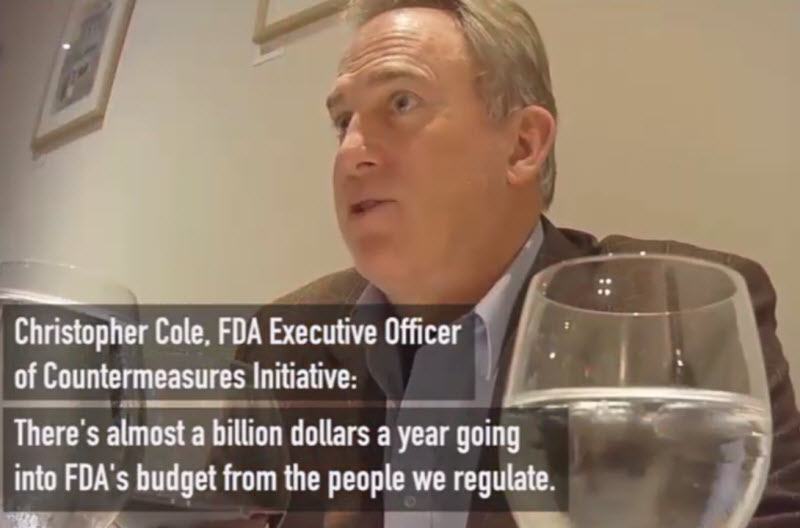
Inner workings of the FDA’s Marriage to Pharma: “Don’t rock the boat”
How did I miss this last year? Project Veritas recorded an FDA executive admitting that the FDA is nothing but a food & pharmaceutical company whore, the approvals are rigged in industries’ favour, and it’s better to “look the other way and not rock the boat” if you want to move up the ranks, and there’s no incentive or protection for people who stand up. Feb. 15, 2022 | Christopher Cole, FDA Exec. Officer on hidden camera.

Let’s help the Vaxxed in 2023 [Jason Christoff #168]
A friend sent this podcast to our telegram group, and I’m so glad she did. It’s really interesting and covers a lot of what we’ve been talking about over the last few years. Mind control, fake-viruses, toxic-shots, graphene, the great reset, 15-minute cities, and the ancient psychos behind all this. We need to help our fellow citizens and kids this year.

[Rant] Satan-Claus Canada #WeWontForget
What will get kids put on the Naughty or Nice List? Well, the “Nice” list includes being masked up, vaccinated, and keeping up-to-date on boosters and flu shots. #NotTodaySatan

“We’re in End Times” Dr. James Thorp (OB/GYN)
An Ob-Gyn for over 40 years talks about the killing fields and the corrupt medical industrial complex murdering the future of humanity.

[Funeral Directors] speaking up about Covid, Boosters & now FLU shots!
Funeral Directors sharing what they are seeing with vaccine deaths, sharing first hand experiences with death certificates and families, and warning not to get the boosters.

[Senator Alex Antic] Injections will go down as greatest medical scandal in history
Senator Alex Antic: “Injections will go down as the greatest medical scandal in history, and none of YOU said a single thing—these injections are harming and in many instances killing our young people, but SA Health keeps pushing the injection narrative.” (21 Nov, 2022)

2 Years to Flatten the Human Spirit
Great conversation between former pharmaceutical rep and two students as they all discuss their awakening during the pandemic.

Are the Kids ok? [C19 Jabs]
Prominent, globally-recognized Cardiologists’ last words before being permanently banned from Twitter

Fired Paediatric Nurse Emotional Testimony “I am the face of your ‘misinformation’ campaign.”
Tawny Buettner, former Californian paediatric nurse who lost her job for asking her supervisors to report adverse effects to the FDA/VAERS, gives an emotional plea at a San Diego Supervisors meeting

Former Pfizer Staff Speak Out
Collection of Former Pfizer Staff and what they have to say. “I feel like I work for an Evil Corporation”. If these ex-and current Pfizer employees are speaking the truth, let’s investigate and figure out what we need to do about it.

Chasing down vaccinators who harmed loved ones with Covid Vaccines
Will we see this more and more as people realize the truth about the dangers of these shots?

Australian Jab Update – Miscarriages & Serious Injuries
Quickie but important Jab Updates for Australians:
(1) TGA FOI 3727 – Pfizer injury data.
(2) Australian gynaecologist Dr. Luke McLinden found that (50% – 74%) had miscarriages after taking the jab.

Sean Hartman’s dad wants to know “What happened to my son?” after Pfizer shot
Sean Hartman’s Dad wants answers to his son’s Pfizer death. Testimony at Toronto City Council board of health. I’m not an anti-vaxxer at all, I’m really not, but I think there is something wrong with this one.

Safe & Effective = Reckless & Dangerous | Julian Gillespie
Lawyer Julian Gillespie is standing up to Australia’s Secretary of Health in Federal Court against the dangerous and deadly shots, warns of big pharma’s influence over politicians and medical authorities, and new oppressive powers being legislated, and calls out the Australian government for ignoring the Pandemic Plan and letting WHO go rogue on the Australian people.

Dr Duncan Syme | Health & Govt bureaucracies have trashed principles
Doctor that was suspended by AHPRA for providing medical exemptions for the experimental gene-therapy injections explains how government bureaucrats have committed atrocities during the pandemic and they want even more power. A call to arms for other fellow health professionals, politicians, the media and the general public to stand up before unprecedented new powers control us all.

Site Notifications/Chat:
- Telegram Post Updates @JourneyToABetterLife (channel)
- Telegram Chatroom @JourneyBetterLifeCHAT (say hi / share info)
- Gettr Post Updates @chesaus (like fakebook)
Videos:




![BlueTRUTH [Part-2/5] Testing “Vaccinated”](https://pennybutler.com/wp-content/uploads/2023/04/BlueTruth-PartTwo-TestingVaccinated.jpg)